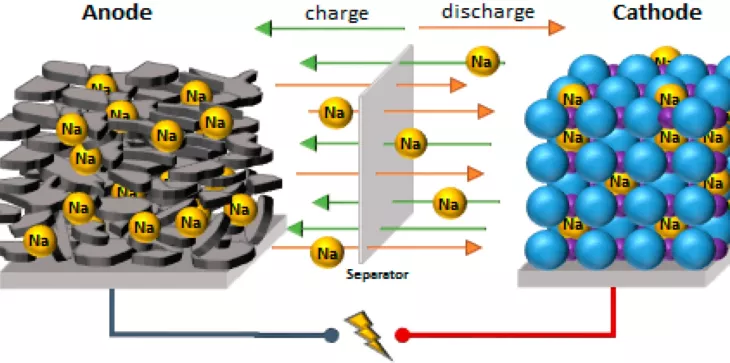
Research carried out in Japan has been able to create a new, highly porous carbon-based material that replaces graphite, and which can be used as an anode in sodium-ion batteries, increasing its energy density above lithium-ion batteries.
A team of researchers from the University of Tokyo has managed to increase the energy density of sodium batteries. It is an energy-efficient method of producing new carbon-based material with a very high sodium storage capacity. The technology is based on taking advantage of the highly porous nanostructure of this charcoal to use it as an anode electrode for the batteries.
The lithium-ion batteries are used today in almost all laptops or electric vehicles. Their advantage is in the energy density they offer, which translates into more autonomy. Their biggest drawback is that the materials they are made of, such as cobalt and lithium, are scarce and expensive. As demand for electric vehicles increases, these materials will become more difficult to obtain and possibly higher in price.
The sodium is cheap, abundant and sustainable, making it a great candidate for energy storage on a large scale. Sodium is the sixth most abundant element on earth. Unfortunately, these batteries do not have as much energy as lithium-ion batteries and also cause degradation problems when it comes to charging and discharging.
The University of Tokyo study published by Angewandte Chemie International Edition details an energy-efficient method to produce a new carbon-based material that increases the storage capacity of sodium-ion batteries.
The objective was to achieve a material for the negative battery electrode that is different from the graphite currently used in lithium-ion batteries. By using magnesium oxide it is possible to adjust the nanostructure of this highly porous material. After multiple experimental and theoretical analyzes, the optimal manufacturing conditions and the ingredients necessary for their production were established, so that it is possible to reach a capacity of 478 mAh / g, the highest for graphite is just 372 mAh / g.
The study will continue to analyze other fundamental parameters of these sodium-ion batteries, such as input and output power, useful life or low temperature operating capacity.